Non-invasive measurement of nuclear relative stiffness from quantitative analysis of microscopy data
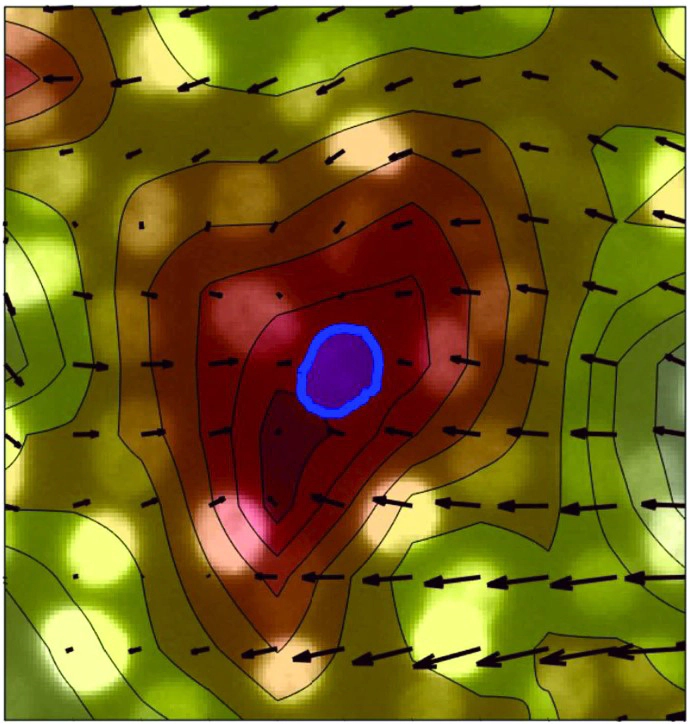 Image credit: Springer
Image credit: SpringerAbstract
The connection between the properties of a cell tissue and those of the single constituent cells remains to be elucidated. At the purely mechanical level, the degree of rigidity of different cellular components, such as the nucleus and the cytoplasm, modulates the interplay between the cell inner processes and the external environment, while simultaneously mediating the mechanical interactions between neighboring cells. Being able to quantify the correlation between single-cell and tissue properties would improve our mechanobiological understanding of cell tissues. Here we develop a methodology to quantitatively extract a set of structural and motility parameters from the analysis of time-lapse movies of nuclei belonging to jammed and flocking cell monolayers. We then study in detail the correlation between the dynamical state of the tissue and the deformation of the nuclei. We observe that the nuclear deformation rate linearly correlates with the local divergence of the velocity field, which leads to a non-invasive estimate of the elastic modulus of the nucleus relative to the one of the cytoplasm. We also find that nuclei belonging to flocking monolayers, subjected to larger mechanical perturbations, are about two time stiffer than nuclei belonging to dynamically arrested monolayers, in agreement with atomic force microscopy results. Our results demonstrate a non-invasive route to the determination of nuclear relative stiffness for cells in a monolayer.